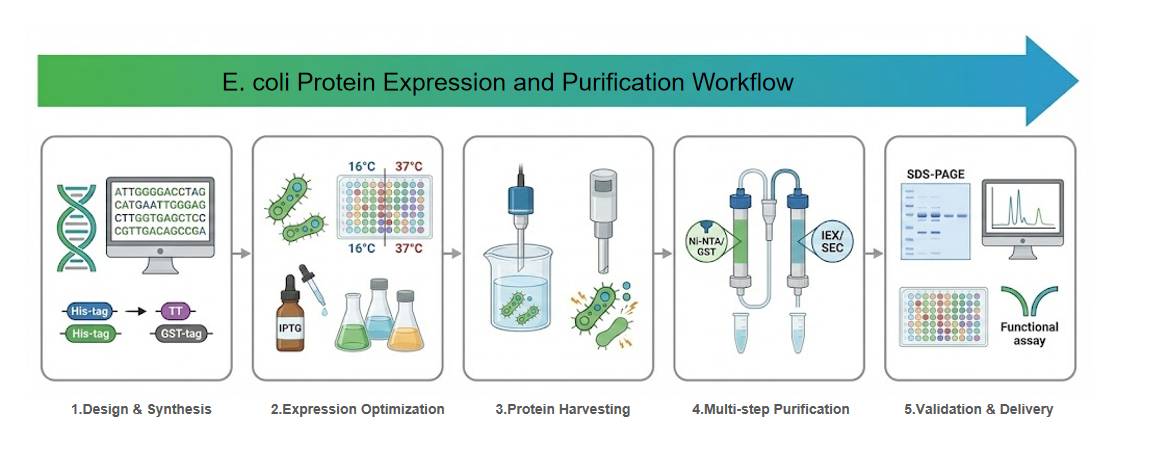
Application Study 1: Scalable HTP Screening for Industrial Expression
To address the "strain selection" bottleneck common in industrial production, academic research has established scalable high-throughput screening platforms. By utilizing Ligation-Independent Cloning (LIC) for rapid library construction and screening diverse E. coli hosts (e.g., BL21(DE3), Rosetta) in 96-well formats, it is possible to rapidly lock in the optimal strain and induction parameters (temperature, time, IPTG) to maximize recombinant yield.
(Reference: Morão LG et al., 2021)
Application Study 2: Managing Challenges with Inclusion Bodies
When expressing eukaryotic proteins, misfolding often leads to insoluble inclusion bodies. Technical literature suggests that low-temperature induction (e.g., 16C) can slow translation rates to promote correct folding. Furthermore, adding chemical chaperones (e.g., glycerol, proline) helps maintain solubility. For proteins already in inclusion bodies, high-purity functional proteins can still be recovered through scientific refolding processes (e.g., removing denaturants while maintaining correct disulfide bond formation).
(Reference: Bhatwa A et al., 2021)
Application Study 3: Specific Protein Expression for High-Sensitivity Diagnostics
In the development of high-sensitivity serological assays, targeting critical protein domains (such as the C-terminal domain of the SARS-CoV-2 S1 subunit) rather than the full-length protein can be a highly effective strategy. By combining affinity chromatography with ion-exchange chromatography for secondary purification and strictly removing endotoxins, recombinant proteins can be obtained that demonstrate excellent antibody-capture capabilities in ELISA tests.
(Reference: Pambudi S et al., 2024)
Application Study 4: Efficient Purification with Column Regeneration
To support large-scale production, research has proposed practical strategies for the regeneration of purification columns. By using specific low-pH or high-salt buffers to clean silica-based columns, it is possible to maintain purity and binding capacity across multiple cycles of reuse, significantly optimizing the costs of downstream purification for industrial-scale projects.
(Reference: Zhou Y et al., 2018)