Raspberry ketone is the main aromatic compound of red raspberry, which has wide applications in cosmetics, food, and medicine. In recent years, raspberry ketone has attracted wide attention as a essence and dietary supplement.
However, the natural abundance of raspberry ketone is low, with only 1-4 milligrams extracted from 1 kilogram of raspberry, making raspberry ketone one of the most expensive natural seasoning ingredients, with a price of up to $20000 per kilogram. The synthesis of raspberry ketone from chemical intermediates through various methods in industry can reduce the cost to several dollars per pound, ranging from one-fifth of the cost of natural products. However, in recent years, due to increased public awareness, the demand for sustainably produced flavor compounds from natural sources has been increasing. The production of raspberry ketone using synthetic biology technology has become a focus of attention.
But even using biosynthesis, the yield of raspberry ketone is not ideal.
Recently, researchers from the Finnish National Center for Technical Research (VTT) synthesized raspberry ketone (30) for the first time in Bensom tobacco using transient expression technology through fusion pathway genes μ G/g) and de novo synthesis of raspberry ketone in brewing yeast, increasing the yield to (60mg/L), which is currently the highest reported yield.
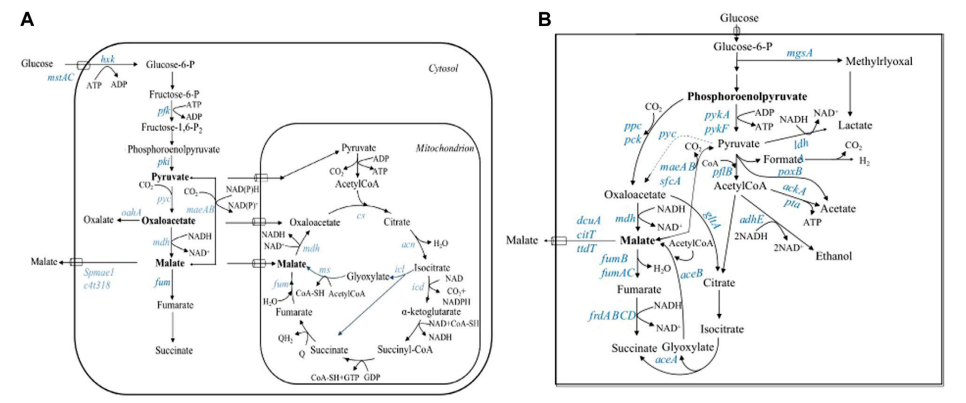
Fig1 Saccharomyces cerevisiae efficiently produces the spice raspberry ketone on a chassis
The intermediate product of the phenylpropanoid pathway, coumarin coenzyme A (CoA), is a direct precursor of raspberry ketone and an important precursor for flavonoid and lignin biosynthesis in plants, thus being highly regulated.
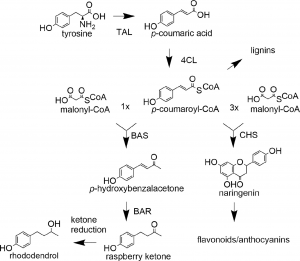
Fig2 The enzymes involved in biosynthesis, raspberry ketone, and the utilization pathways of coumarin CoA in other aspects
Researchers have found that the synthetic fusion of 4-coumaric acid ligase (4CL) and benzoacetone synthase (BAS) (hereinafter referred to as 4CL-BAS) can transport coumaric acid CoA from the ligase channel to the synthase. 4CL-BAS using natural BAS can achieve high yield in Bensom tobacco, and its effect is superior to that of BAS mutase. In this study, for the first time, the highest yield of raspberry ketone expressed instantaneously in Bensom tobacco exceeded 30 μ G/g.
In brewing yeast, using p-HCA (coumarin acid) as a precursor and 4CL-BAS from natural BAS, a maximum of 61mg/L raspberry ketone can be produced in an optimized production system.
P-HCA is generated through a two-step enzymatic reaction using PAL (phenylalanine ammonium lyase) and C4H (cytochrome P450 dependent monooxygenase cinnamate 4-hydroxylase) from phenylalanine, or through a single enzymatic reaction using tyrosine through TAL. In the study, tyrosine was used as the substrate to produce p-HCA using TAL (tyrosine ammonium lyase), achieving de novo synthesis of raspberry ketone with a maximum yield of 47 mg/L.
The author pointed out in the article that both values (61mg/L and 47mg/L) indicate the highest level of raspberry ketone content in the yeast system.
The synthesis pathway and industrial applications of raspberry ketone using yeast as a fermentation medium.
Raspberry ketone, a popular flavoring agent, is synthesized through a specific pathway using yeast as the fermentation medium. The production process is as follows:
1. Yeast culture: Cultivate a specific strain of yeast (e.g., Saccharomyces cerevisiae) in a suitable growth medium.
2. Fermentation: Inoculate the yeast culture in a fermentation vessel containing the necessary nutrients, such as sugars and salts. The fermentation process allows the yeast to convert the available sugars into alcohol, generating ethanol as a byproduct.
3. Bioconversion: Once the fermentation is complete, extract the ethanol from the fermented broth. This step involves distillation and other purification techniques to obtain a high-purity ethanol solution.
4. Ketone synthesis: Use the ethanol solution as a reactant in the chemical synthesis of raspberry ketone. Various methods can be employed, including oxidation reactions or direct ketone formation from ethanol.
5. Purification: Purify the synthesized raspberry ketone to remove impurities and obtain a high-quality product. This may involve techniques such as distillation, crystallization, or chromatography.
Industrial applications:
Raspberry ketone finds widespread use in various industries, notably in the food, cosmetic, and pharmaceutical sectors. Some key industrial applications include:
1. Flavoring agent: Raspberry ketone is used as a natural flavoring agent in food and beverages, providing a characteristic fruity and aromatic taste. It is commonly used in confectionery, baked goods, dairy products, and beverages.
2. Fragrance ingredient: Due to its pleasant aroma, raspberry ketone is utilized as a fragrance ingredient in perfumes, cosmetics, and personal care products. It adds a fruity, sweet scent to these products.
3. Weight loss and dietary supplements: Raspberry ketone is marketed as a weight loss and dietary supplement due to its purported ability.
CD Biosynsis has established a powerful synthetic biology platform to integrate engineering principles and a broad range of methodologies from various disciplines, including biology, bioinformatics, chemistry, physics, mathematics, and computer science.